| |
NeuroStar Advanced Therapy TMS
FDA-Approved since 2008, with over 2 million successful treatments delivered. Transcranial Magnetic Stimulation (TMS) uses short targeted pulses of magnetic energy to stimulate nerve cells in the brain. This happens while the patient is awake and alert, the treatment stimulates areas of the brain that are underactive in depression.
Due to its innovative and proven success, TMS therapy is covered by most insurance plans.
| |
How TMS Works:
Learn what makes TMS something different and why you should consider this for treatment of depression
| |
TMS is proven effective with a majority of patients experiencing relief within several weeks
TMS provides long lasting symptom relief that is targeted at the source
Non-Invasive and Drug Free
TMS utilizes a non-invasive magnetic field that activates the natural function of the brain. As a result, it is medication free with virtually no side effects
| |
What to Expect:
Find out how simple and easy TMS treatment can be.
| |
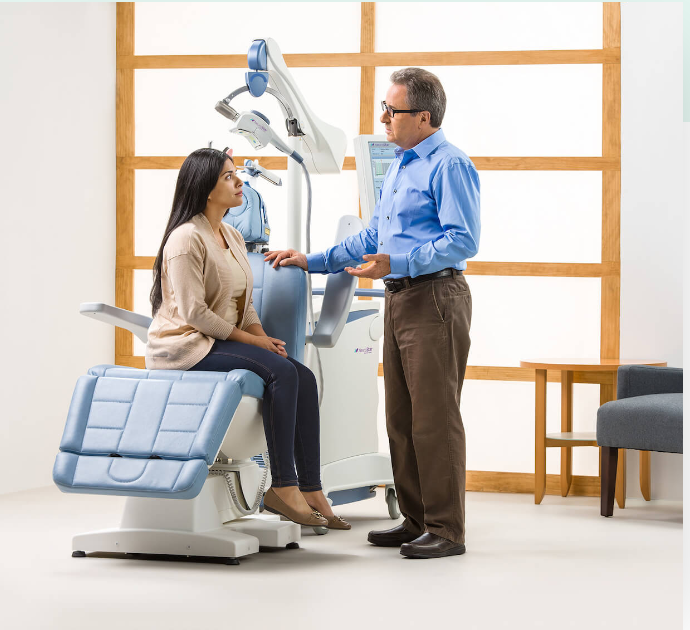
You will recline comfortably in the TMS treatment chair. A small, curved device containing the magnetic coil will be positioned lightly on your head.
The coil delivers focused magnetic stimulation directly to the target areas of the brain. You will hear a clicking sound and feel a tapping sensation on your head.
After Treatment
You can resume your normal activities immediately. Because there are no effects on alertness or memory, you can drive yourself to and from treatment sessions. You will remain awake and alert during treatment with no sedation or anesthesia.
TMS is a highly focused MRI-Strength magnetic pulse that non-invasively stimulates the brain and treats major depression.
Specifically: The magnetic pulse depolarizes the cortical neurons in the brain which promotes the release of neurotransmitters. By depolarizing these neurons it modulates deeper regions of the brain that effect mood. Repeated pulse stimulation to this region of the brain are proven to have anti-depressant effect. TMS therapy eliminates the side effects and adverse events often associated with antidepressant medications because it does not circulate throughout the body. This targeted approach to treatment is FDA approved, safe and effective.
Unlike ECT, there are no negative long-term effects associated with TMS therapy. TMS therapy continues to gain popularity with physicians and patients because it is safe, effective and tolerable.
Discontinuing your medication is a decision that needs to be made between you and your healthcare provider. However, TMS therapy can be administered while a patient is on medication. It is actually preferred that you remain on your medication until therapy has completed.
Patients have reported feeling differently as early as day 1. Some patients don't feel anything at all until around the 4-week mark. The response will be individual to you as a patient.
Each treatment session lasts anywhere from 18 to 30minutes depending on the prescribed dose. The initial course of treatment will involve at least 30 consecutive treatment sessions over a 6-week period. That is a treatment session each day Monday thru Friday, plus 6 taper sessions in week 7 and 8.
All patients respond differently to TMS therapy. However, clinical studies support that 45% of patients achieve full remission from major depressive symptoms. That's nearly half of patients going on to live their life free from depression. 68% of patients respond positively to TMS therapy. Response is classified as a 50% reduction in symptoms from baseline. That is nearly 7/10 patients that go on to live their lives with less depression and a higher quality life. There have been over 2.5million treatments with TMS with the numbers continuing to rapidly grow as more patients are considering TMS as a treatment option.
Patients report feeling a light tap on the top of their head underneath the treatment coil. This tapping sensation is a result of the magnet delivering pulses for treatment. Few patients have reported scalp discomfort at the treatment site.